Wim Hof Has Less Brown Fat Than You Do. That Matters for Cancer Research.
April 13, 2026
A look into our Wim Hof Method (WHM) as novel therapy for cancer open science feasibility study. The development of the first multimodal dataset linking breathwork, cold exposure, inflammation, and tumor metabolism, and what AI can do with it that nothing else can.
Jim Nasr, Co-Investigator · WHM for Cancer Open Science Project · coldtherapy.acoer.com
Wim Hof has less brown fat than most people do.
That statement is not theatre. In 2018, Muzik and colleagues published a PET-CT and fMRI study in NeuroImage showing that Wim Hof's extraordinary cold tolerance, as many may have suspected, is not driven by brown adipose tissue (BAT) at all.[5] The dominant metabolic signal came from somewhere else entirely: forceful activation of the intercostal muscles during his breathing protocol, producing heat that dissipates into adjacent lung tissue and warms blood in the pulmonary capillaries. The man with the global folklore for cold tolerance was shown to have less BAT than a random tested adult.[5,6]
Why does this matter for cancer research?
In 2022, a group at the Karolinska Institutet published findings in Nature showed that cold-activated (at 4°C) BAT suppressed tumor growth by up to 80% across multiple cancer models in mice, with a preliminary signal reproduced in a single Hodgkin's lymphoma human patient exposed to 22°C for one week.[1] The mechanism is not cold killing cancer. It is metabolic competition. BAT burns glucose to make heat, tumors run on glucose, and if you push enough glucose toward BAT there is not enough left to fuel the tumor.
That framing is right, and useful. But if BAT is the whole story, how does Wim Hof do what he does? And the more interesting question: if his breathing protocol engages a separate, voluntary, trainable thermogenic pathway on top of whatever BAT contributes, could that pathway be doing real metabolic work against tumors that nobody has ever actually measured?
Our team is running one of the first open science feasibility studies designed to find out.
We are looking at whether the Wim Hof Method, breathwork plus cold exposure plus committed practice, can be safely used by adults living with cancer, and whether it generates measurable signals in inflammation, metabolism, and autonomic function.
The protocol is a classical clinical study in many respects. Our vision for data insights is not.
The spine of this story is about the data. Specifically: what becomes possible when you combine a multimodal longitudinal participant-owned dataset, a 26-study evidence base we have already harmonized and made query-able, and AI methods that did not exist the last time anyone tried to study breathwork and cancer seriously.
Tumors are greedy. Cold exposure can out-compete them for glucose.
Otto Warburg, the German biochemist and Nobel laureate, described the core metabolic vulnerability nearly a century ago. Cancer cells prefer aerobic glycolysis even when they have plenty of oxygen to do otherwise. That preference is their strength, and it is their liability. Any intervention that meaningfully shifts circulating glucose away from the tumor microenvironment has, at minimum, theoretical anti-tumor potential.
Brown adipose tissue (BAT) is one of the cleanest ways to do that. BAT is a thermogenic organ. Its primary function is non-shivering heat generation through mitochondrial uncoupling protein 1 (UCP1). When activated by cold, it consumes significant quantities of glucose and lipids to produce heat. In the Karolinska mouse study, cold-exposed animals showed dramatic glucose redistribution. Uptake was concentrated in BAT and nearly undetectable in tumor tissue. GLUT1 transporters in tumors dropped. The PI3K/AKT/mTOR signaling axis in tumors was suppressed while the same axis in BAT was upregulated.[1,2]
The causal proof came from the knock-out experiments. Remove the BAT surgically, or feed the mice a high-glucose diet during cold exposure, and the anti-tumor effect disappears.[1] The mechanism is metabolic competition, not a cytotoxic effect of cold temperature on the tumor.
This is consistent with what we already know about glucose restriction as a therapeutic strategy. Caloric restriction, ketogenic diets, and pharmacological glucose-lowering drugs work on the same principle. The distinguishing feature of cold-induced BAT activation is that it does not require dietary modification or medication. People can do it at home. That has always been the argument for its clinical appeal.
BAT is not the only thermogenic engine. The other one is voluntary.
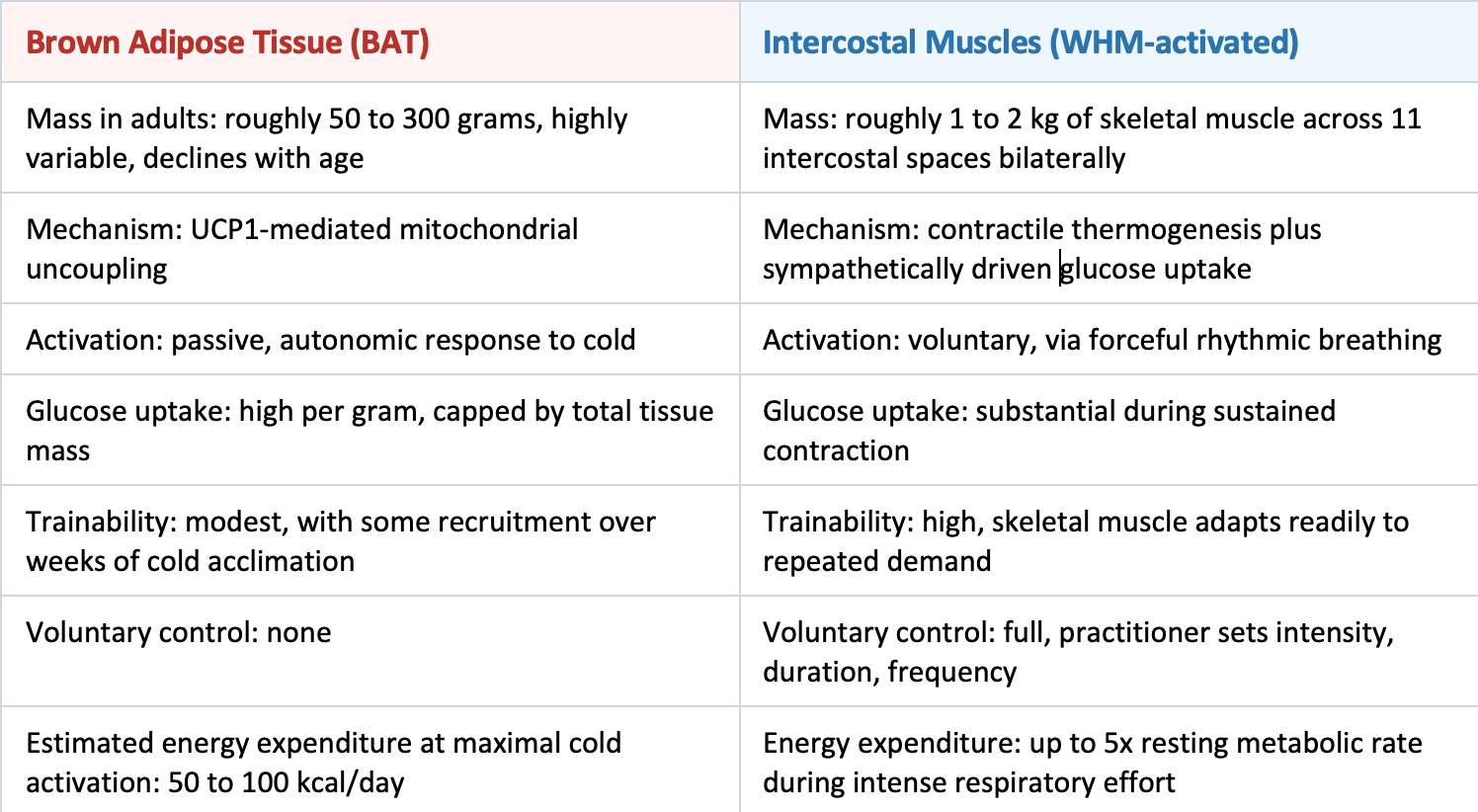
The scientific literature on cold-induced thermogenesis has focused almost exclusively on brown fat. That is not wrong. It is simply incomplete. The Muzik study is the clearest signal we have that something else is doing serious work, and that something else is a group of muscles nobody has been looking at.[5]
Muzik's team expected to see the standard pattern: elevated glucose uptake in BAT, straightforward cold-induced activation. They did not get that. The dominant metabolic signal came from the intercostal muscles. Forceful WHM breathing drove sympathetic innervation and glucose consumption in the muscles between the ribs. The heat produced dissipated into the adjacent lung tissue and warmed circulating blood in the pulmonary capillaries. And Wim Hof himself was shown to have less BAT than an average person off the street.[5,6]
That finding should have changed the conversation. It mostly did not. It says, quietly, that Hof's physiology is not powered by a large brown fat depot. It is powered by a voluntarily trained thermogenic pathway in muscles he has been activating every day for decades.
We recorded a conversation with Wim Hof recently (see embedded video) where he made this point in his own words. The intercostals, in his view, are not accessory breathing muscles. They are a trainable metabolic organ. A full session of the breathing method (typically three to four rounds of 30 to 40 forceful breath cycles with retention holds) engages them hard enough to generate more heat than brown fat alone could produce. We are not aware of any clinical study that has properly measured this.
The comparative biology
Here is why that distinction matters for cancer metabolism.
If the dominant glucose-burning engine during WHM practice is the intercostals and not BAT, three things change:
First, glucose diversion becomes controllable. The practitioner can dial the intensity up or down by adjusting the breathing protocol. Brown fat cannot be consciously engaged. Intercostal muscles can.
Second, trainability. BAT declines with age and with increased body fat, both of which correlate with cancer incidence. Skeletal muscle adapts at any age. A 55-year-old cancer patient can train their intercostals. They cannot grow a new brown fat depot on command.
Third, and most important for the cancer question: skeletal muscle is already the body's largest glucose sink. It accounts for roughly 80% of insulin-stimulated glucose disposal.[16] Even modest sustained activation of the intercostals may produce a glucose sink large enough to compete with tumor metabolism on its own.
Intercostals from the breathing protocol. Nobody has ever quantified the combined glucose demand of that dual activation in a clinical setting. Doing so is one of the central scientific opportunities of our study.
THE UNANSWERED QUESTION
If the breathing alone can create meaningful metabolic competition with tumors, independent of the cold exposure, we should know about it. Nobody has tested it in a cancer population. Our feasibility study will collect the breathing session data (rounds, retention times, frequency) alongside the metabolic markers (fasting glucose, insulin, HOMA-IR, hs-CRP) needed to explore the question for the first time.
26 studies, 20 years, one harmonized dataset.
Our work does not start from scratch. Before we wrote our open science protocol, we curated and normalized 26 peer-reviewed studies on cold exposure and human metabolism spanning 2002 to 2022. The result is publicly available through the Cold Therapy Evidence Explorer. It is the analytical substrate on which every downstream decision in the study rests: the outcome measures, the biomarker shortlist, the AI model design.

The measurements captured across these 26 studies are unusually rich. Energy expenditure at every level, total, daytime, sleeping, and resting metabolic rate, all corrected for fat-free mass. BAT activation via PET-CT. Respiratory quotient, VO2 and VCO2, heart rate, core and skin temperature with proximal, distal, and gradient calculations. Blood markers including plasma glucose, insulin, norepinephrine, epinephrine, free fatty acids, glycerol, leptin, ghrelin, adiponectin, triglycerides, TSH, freeT3, free T4, cortisol, and ACTH. The research groups span Maastricht University, the Karolinska Institutet, Tenshi College in Japan, the University of Sherbrooke in Canada, and institutions across the Netherlands, Finland, Switzerland, Singapore, Spain, Australia, Sweden, and the US.
Our initial work was not just the curation. It was the harmonization. We normalized these heterogeneous studies into a common schema: cold temperature, control temperature, exposure duration, participant profile, BAT activation status, metabolic outcome. That level of cross-study comparison is impossible from reading the individual papers, and it is exactly what let us make specific, evidence-grounded choices about our own protocol. Progressive cold temperatures from ambient down to 10 to 16°C. The biomarker shortlist (hs-CRP, IL-6, fasting glucose, insulin). The wearable metrics we collect (HRV, resting heart rate, body temperature). The full dataset and interactive dashboards are at coldtherapy.acoer.com/dashboard.
Small pilot. Significant dataset.
Our feasibility pilot is deliberately small: 15 to 20 participants in our treatment group, 16 weeks, fully remote. The architecture of data it will generate is not. As far as we know, this is the first cancer cohort that will produce all of the following streams at once, longitudinally, under a standardized WHM protocol:
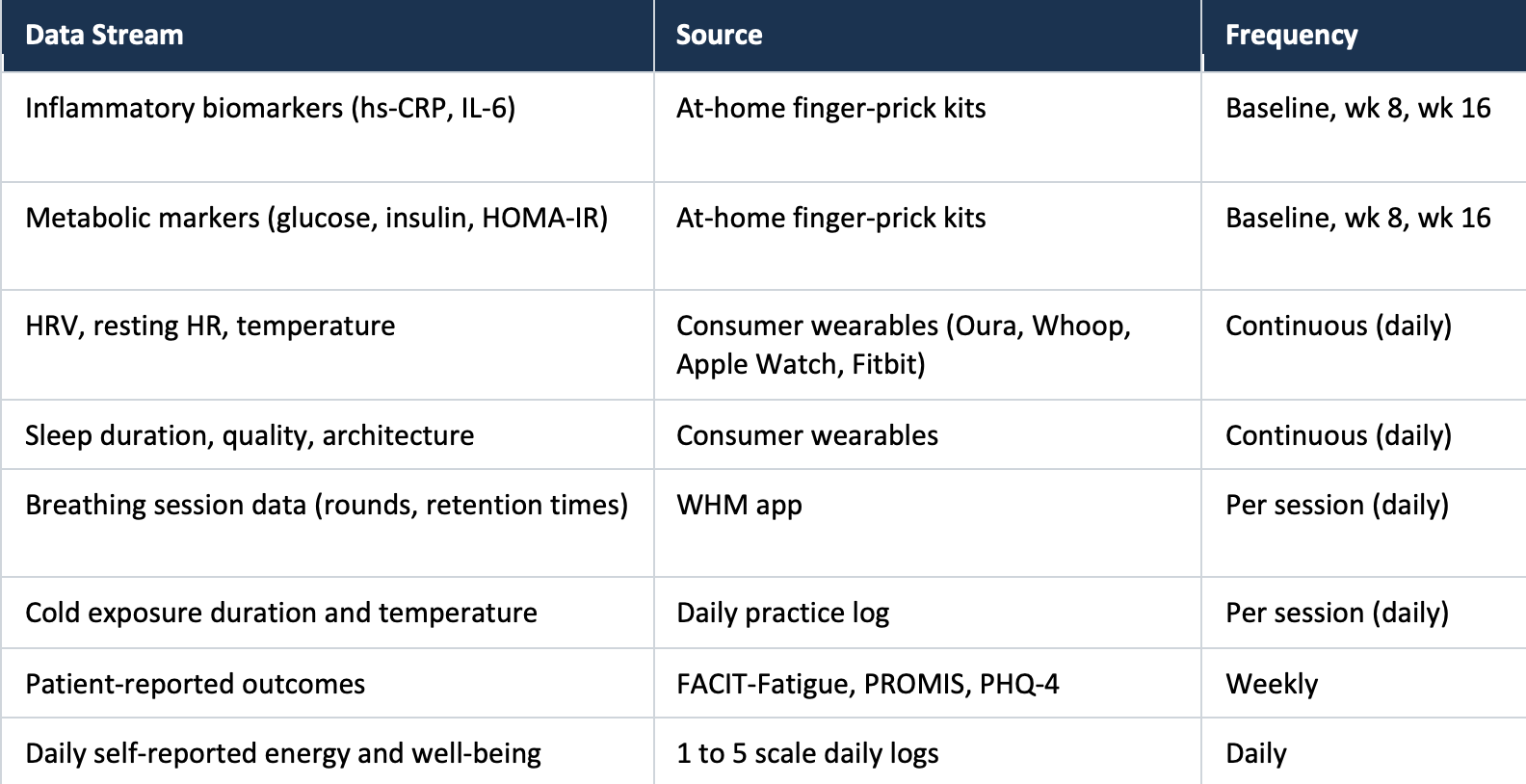
For a 16-week study with 20 participants, that design produces roughly 2,240 daily observation vectors. Each vector carries wearable biometrics, practice metrics, and self-reported outcomes, alongside three waves of blood-based biomarkers and weekly validated psychometric instruments. That is an unusually dense temporal dataset for a cancer cohort. The density is what makes the advanced computational methods worth attempting at all.
Every participant owns their data. Consent is recorded on-chain and revocable at any time. Every data input and every AI training operation is cryptographically verified on a public blockchain, so the provenance of every analytical claim can be independently audited.[7]
We recognize that lingo sounds pitch deck-y. However, it’s anything but in practice. Most clinical research still treats participant data as something the sponsor holds and the patient borrows. We think it should be the other way around. Data sovereignty should not be a premium feature of open science. It should be the baseline.
Our plans with AI, and why it’s beyond anything done before.
For our study, we’ll be using traditional statistics to still do the heavy lifting on the feasibility question itself. Paired t-tests, Wilcoxon signed-rank, mixed-effects models. That is the responsible floor for a study this size. However, the dataset we are building is structured to support a much more ambitious AI agenda.
Five ways we plan to use AI to go well beyond statistical methods:
1. Causal machine learning, not just correlation
The hardest thing in any single-arm observational study is separating causal effect from confounding. We plan to apply causal forests and double machine learning to estimate whether adherence to the protocol has an identifiable causal effect on outcomes like fatigue, HRV, and hs-CRP, after adjusting for the confounders we can actually measure. Medication changes. Infections. Cancer progression. Differences between wearable devices.
Causal forests (Wager, S., & Athey, S. 2018), handle high-dimensional correlated data well, pick up non-linear effects, and are doubly robust to observed confounders.[8] They can estimate average treatment effects and, more usefully, conditional average treatment effects for specific subgroups, without forcing a parametric model on the data. In a heterogeneous cancer cohort, the ability to detect who responds is more scientifically useful than a single average effect. Cancer patients are not interchangeable. The method should not treat them as such.
Novel Proposal: Causal Dose-Response Curves for Cold Exposure and Breathing Intensity
We will use recent neural network methods for estimating individual dose-response curves from observational data. The goal is to model the continuous relationship between cold exposure intensity (duration x temperature x frequency) and breathing intensity (rounds x retention time x session frequency) against downstream outcomes (inflammation, HRV, fatigue, glucose) at the participant level, not the group level. Given the Muzik findings on intercostal thermogenesis, separating the dose-response of the breathing from the cold exposure is scientifically critical. Nobody has done it in any cancer population.
2. Digital twins that actually know the practice
A digital twin in medicine is a computational replica of an individual patient, continuously updated with real data, that can simulate what happens next and evaluate hypothetical interventions in silico. In 2025, researchers showed that large language model architectures can be adapted for clinical trajectory prediction. DT-GPT (Digital Twin Generative Pretrained Transformer) outperformed conventional ML on non-small-cell lung cancer data.[9]
Our pilot produces exactly the kind of data a digital twin architecture needs to be useful. Each participant generates more than 100 days of dense multi-dimensional time series. Not enough to train a foundation model from scratch. Enough to begin fine-tuning personalized trajectory models.
Novel Proposal: WHM Digital Twins for Cancer Patients
We propose building a personalized computational model for each participant. The model integrates their wearable biometrics, breathing practice data, cold exposure logs, biomarker trajectories, and patient-reported outcomes into a unified probabilistic representation. Three things become possible: (1) Real-time prediction of biomarker trajectories given current practice patterns. (2) Counterfactual simulation ("what would have happened if breathing intensity had increased at week 8?"). (3) Protocol optimization ("what combination of rounds, retention duration, and cold exposure maximizes HRV improvement for this individual?"). As far as we can tell, nobody has applied digital twin methods to a breathwork and cold exposure intervention in oncology. This would be the first.
3. Multimodal foundation models and cross-modal attention
One of the genuinely useful developments in biomedical AI over the last few years is the rise of multimodal foundation models. These are large neural networks pretrained on diverse data types that learn transferable representations across tasks.[10] Our dataset spans several modalities: continuous physiological signals, structured behavioral logs, ordinal psychometric data, and interval-sampled serum biomarkers. That heterogeneity is what these models are built for.
Novel Proposal: Cross-Modal Attention for Practice-Biomarker Coupling
Train a transformer-based attention model on the aligned multimodal time-series to discover which specific practice patterns attend most strongly to downstream biomarker changes. The attention weights themselves become interpretable scientific hypotheses. For example: the model might find that morning breath retention times above 90 seconds, when followed by cold exposure within 30 minutes, are strongly predictive of HRV improvement 48 hours later. No human analyst would find that temporal coupling pattern across 20 participants and 112 days of multimodal data. A well-trained attention mechanism might.
4. A knowledge graph that updates with every new participant
A knowledge graph is a structured, query-able representation of every entity and relationship in a domain, carrying directionality, strength, and provenance. Unlike a static literature review, a knowledge graph can be reasoned over. Questions become path queries. New data updates edge weights in place instead of forcing the whole thing to be rewritten.
The visualization below represents a portion of the WHM-Cancer knowledge graph we will construct from the convergence of our 26-study evidence base, the pilot data, and the broader literature.
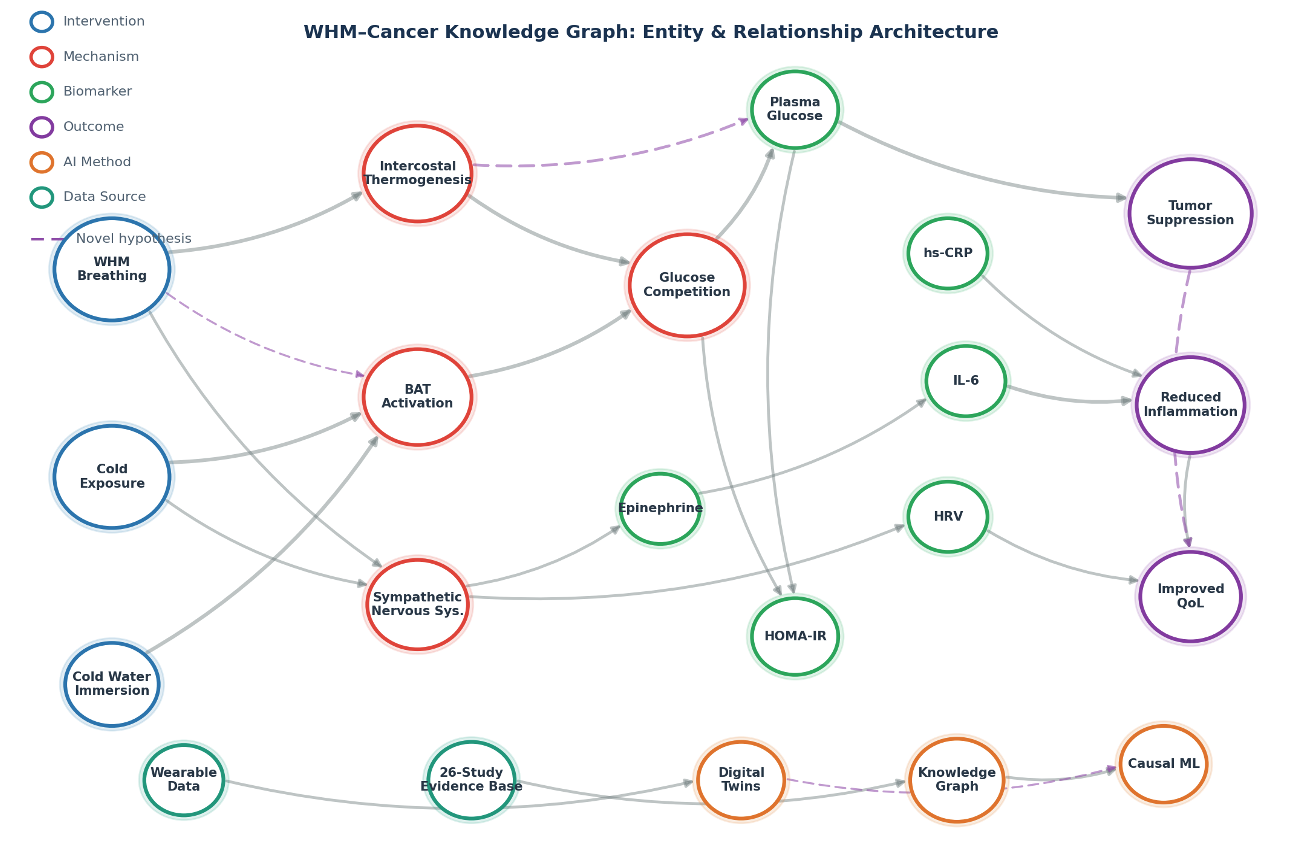
The graph encodes relationships our evidence base has already established (cold exposure → BAT activation → glucose uptake → tumor suppression) along side the novel hypotheses the pilot will actually test (WHM breathing → intercostal thermogenesis → glucose competition). Apply a graph neural network to that structure and you can identify inference paths: multi-hop chains of evidence linking interventions to outcomes through intermediate mechanisms. No single paper has ever tested those paths end to end. A query might surface: "WHM breathing → intercostal activation → increased glucose disposal → reduced plasma glucose → reduced HOMA-IR → reduced IL-6 → tumor metabolic stress." Each edge has a confidence weight drawn from the evidence base. New pilot data moves those weights in real time.
Novel Proposal: Graph-Augmented Hypothesis Generation
Train a graph neural network on the WHM-Cancer knowledge graph to do three things. Predict missing edges ("does intercostal thermogenesis have a direct relationship with IL-6 reduction?"). Rank intervention combinations by their predicted effect on target outcomes via path analysis. Identify the highest-value next measurements by computing which unobserved edges would most reduce uncertainty in the graph. This is active learning on the knowledge graph. It would let the AI help design the next experiment: which biomarkers to add, which practice variables to standardize, which subgroups to recruit for a follow-on RCT.
5. Phenotype discovery, and a federated model participants actually own
Not every cancer patient will respond to WHM the same way. Using dynamic time warping plus hierarchical clustering on aligned streams of HRV, inflammation, sleep quality, and adherence, we can ask whether participants separate into distinct response trajectories. These findings are hypothesis-generating, not confirmatory. They are exactly the kind of finding that directly informs stratification for the follow-on randomized controlled trial.
Novel Proposal: Participant-Sovereign Federated Intelligence
Host a local model on each participant's own device, trained on their own data. Each local model contributes gradient updates to a shared global model without ever transmitting raw health data. The participant retains cryptographic proof of their contribution via on-chain attestation. Call it participant-owned federated intelligence. The people generating health data become co-owners of the intelligence derived from it.[7,11]
This is where decentralized science (DeSci) and AI converge in the actual build, not just in tweets or conference slides. We think this is the right direction to build open science proactively.
What this study does that most studies don't.
Participant data sovereignty as the default. Participants own their data. Consent is on-chain and revocable. Every analytical operation is verifiable. This is not stewardship bolted onto the end of a study. It is how day one is structured.
Blockchain-verified provenance. Cryptographic fingerprints of datasets are recorded on a public blockchain with immutable timestamps. Anyone can verify data authenticity by comparing published datasets against the chain record.[7] That is the structural antidote to the credibility problem in clinical research: nobody can see the raw workings.
26 studies, harmonized, not cherry-picked. The protocol is grounded in a curated and normalized evidence base surfaced through an interactive Cold Therapy Evidence Explorer. Not a single promising paper. Not an intuition. Rigorous indexing of two decades of converging evidence.
Open science by default. The statistical code, the machine learning pipelines, and the anonymized individual-level data will all be released publicly. Positive findings and null findings will be reported with the same rigor. A null finding in feasibility is still useful, provided it is fully documented.
Decentralized Science & AI: built not just marketed. Transparent, participant-governed, computationally sophisticated, verifiable end to end. Recent literature calls the convergence "DeScAI".[11] We prefer to build it rather than label it.
What this becomes if feasibility holds.
If feasibility is demonstrated, the implications extend well beyond this pilot. The causal dose-response models, digital twins, knowledge graph, and response phenotypes directly inform the design of the follow-on randomized controlled trial. They tell us who to recruit, what protocol to prescribe, what to measure, and how long the trial needs to run. The AI produced by the pilot becomes the design intelligence for the definitive trial.
More broadly, this work contributes to a shift in integrative oncology that is overdue. Lifestyle interventions have been treated for too long as vaguely beneficial adjuncts, too soft for serious measurement.
We are proposing the opposite. If WHM practice can be characterized with the same rigor as a pharmacological intervention, with dose-response curves, responder profiles, and mechanistic biomarker signatures, then it can be evaluated, compared, and prescribed with corresponding clinical confidence. Perhaps, some sacred cows in oncology research have to be put to pasture. The idea that breathwork and cold exposure cannot be measured the same way a drug can be measured is one of them.
Wim Hof has been asking a specific question for decades. Can his method help people who are sick? It deserves a scientific answer. The tools to produce that answer, from multimodal wearable data to causal machine learning to blockchain-verified open science, finally exist. This pilot is where they come together.
AN INVITATION
We are actively recruiting participants for the treatment group. There is no cost to participate. As a participant, you are not a subject. You are one of our co-investigators. If you are living with cancer and want to contribute to this research, visit coldtherapy.acoer.com or reach out through our proposal on ResearchHub.
References
1. Seki Y, Berglund K, Mandrup S, et al. Brown-fat-mediated tumour suppression by cold-altered global metabolism. Nature. 2022;608:421-428.
2. Chen Y, Kang S. Cold snap for cancer: cold-induced brown fat thermogenesis starves tumor growth. Signal Transduction and Targeted Therapy. 2023;8:37.
3. Kox M,van Eijk LT, Zwaag J, et al. Voluntary activation of the sympathetic nervous system and attenuation of the innate immune response in humans. Proc Natl Acad Sci USA. 2014;111(20):7379-7384.
4. Vetter S, et al. Does the Wim Hof Method have a beneficial impact on physiological and psychological outcomes? A systematic review. PLOS ONE. 2024;19(3):e0286933.
5. Muzik O, Reilly KT, Diwadkar VA. Brain over body: a study on the willful regulation of autonomic function during cold exposure. NeuroImage. 2018;172:632-641.
6. Carney S. Forget Wim Hof. Forget Brown Fat. Remember This. scottcarney.com. 2018.
7. DeScAI: the convergence of decentralized science and artificial intelligence. Frontiers in Blockchain. 2025.
8. Athey S, Tibshirani J, Wager S. Generalized random forests. Annals of Statistics. 2019;47(2):1148-1178.
9. Large language models forecast patient health trajectories enabling digital twins. npj Digital Medicine. 2025.
10. Alsaedi A, et al. Beyond digital twins: foundation models in precision medicine. FEBSOpen Bio. 2025.
11. Decentralized Clinical Trials and Patient Data Control: Blockchain Revolution in Healthcare. 2025.
12. Buijze GA, Sierevelt IN, et al. The effect of cold showering on health and work: a randomized controlled trial. PLoS ONE. 2016;11(9):e0161749.
13. Skeletal Muscle Thermogenesis and Its Role in Whole Body Energy Metabolism. Diabetes & Metabolism Journal. 2017.
14. Cold exposure and human metabolism: A heterogeneous response across tissues and organs. Temperature. 2025.
